Paul U Cameron
Doherty Institute for Infection and Immunity, University of Melbourne, Melbourne, VIC
Last reviewed: November 2019
The cellular immune system
The cellular immune system is the main component of adaptive immune responses that target virally infected cells and is preferentially destroyed in HIV disease. This section will highlight aspects of adaptive and innate immunity including T-cell development, antigen presentation and recognition, T-cell memory, chemokine biology, lymph node architecture and B cell development, relevant to HIV disease pathogenesis.
T-cell development
Stem cells continuously migrate from the bone marrow to the thymus. Although the thymus partially involutes with age, T cells continue to develop in the thymus throughout life. (Jamieson et al., 1999; Kumar et al., 2018) T-cell development in the thymus involves processes for both positive and negative selection that result in the production of T cells with the capacity to recognise foreign or non-self antigens (Ag) (Klein et al., 2014) (Figure 1).
Figure 1. Generation of T cells in the thymus
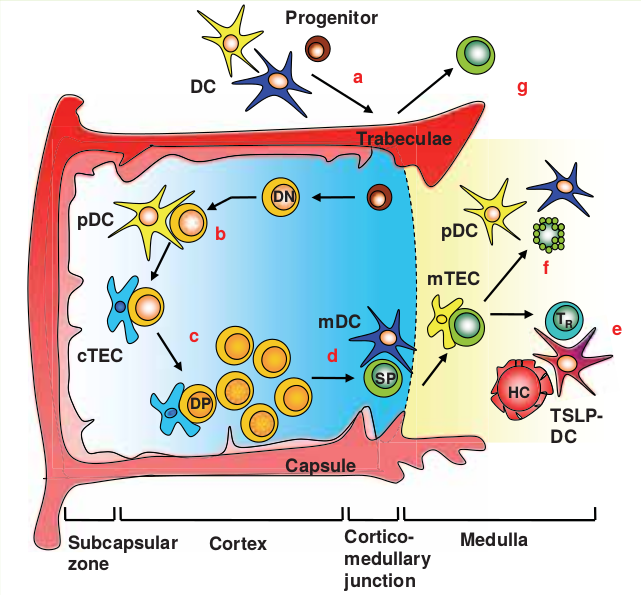
Note: The thymus is divided into lobules separated by trabeculae. Each lobule has a cortex and medulla. T-cell precursors enter the thymus and conditioned naïve T cells exit at the corticomedullary junction (a). The immature double negative (DN; CD3-CD4-CD8-) thymocytes develop and migrate to the cortex where they interact with plasmacytoid dendritic cells (pDC) and cortical thymic epithelial cells (cTEC) (b). Positive selection for low affinity T-cell receptors (TcRs) interacting with self major histocompatibility complex (MHC) molecules and expansion of double positive (DP; CD3+CD4+CD8+) cortical thymocytes occurs in the cortex (c). The positively selected single positive (SP; CD3+CD4+CD8- and CD3+CD4-CD8+) thymocytes migrate to the corticomedullary junction where further interactions with myeloid dendritic cells (mDC) and pDC and medullary thymic epithelial cells (mTEC) lead to negative selection and elimination of high affinity self-reactive T cells (d). Expansion of thymocytes with intermediate affinity for self, into T-regulatory cells (Treg) occurs during interaction of SP thymocytes with DC modified by the cytokine thymic stromal lymphopoietin (TSLP) generated by the Hassall’s corpuscles (HCs) (e). Thymocytes are destroyed during negative selection with DC (f) or survive the conditioning to emerge as recent thymic emigrants (RTE) (g).
Source: Evans VA, Cameron PU, Lewin SR. Human thymic dendritic cells: Regulators of T cell development in health and HIV-1 infection. Clin Immunol 2008; 1126:1-12. Used with permission
In humanised mouse models of HIV infection, reconstitution of the human immune response depends on thymic reconstitution or transplantation (Hatziioannou and Evans, 2012). The relationship between thymic production and subsequent destruction of T cells in HIV infection is summarised in Figure 2.
Figure 2. Pathways for CD4+ T cell depletion in HIV infection
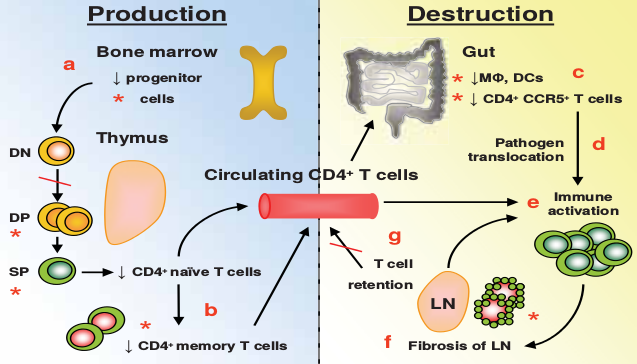
Note: Depletion of CD4+ T cells in HIV infection may occur as a result of direct infection (*) and death of CD4+ T cells or by indirect mechanisms. HIV-1 infection has been associated with impaired production of CD34 progenitor cells in the bone marrow (a), reduced proliferation of thymocytes (Dion et al., 2004) and direct infection of CD4+ thymocytes leading to reduced numbers of recent thymic emigrants and naïve CD4+ T cells. (Su et al., 1995; Bonyhadi et al., 1993) In addition, HIV-1 directly infects circulating CD4+ memory T cells although at low frequency (b). At the same time, there is significant depletion of mucosal CD4+ T cells (by direct infection of both CCR5+ and CCR5- CD4+ T cells), DCs and macrophages (MΦ) (c). (Brenchley et al., 2004) This may potentially compromise the integrity of the mucosal barrier leading to increased translocation of bacteria from the intestinal lumen (d). (Brenchley et al., 2006b) High levels of immune activation (e) increase the proliferation and death of both CD4+ and CD8+ T cells, which are associated with lymph node (LN) fibrosis (f ) and retention of T cells in the LN (g) immune activation (e) increase the proliferation and death of both CD4+ and CD8+ T cells, which are associated with lymph node (LN) fibrosis (f ) and retention of T cells in the LN (g). (Brenchley et al., 2006a) DN=double negative; DP=double positive; SP=single positive thymocytes.
Source: Evans VA, Cameron PU, Lewin SR. Human thymic dendritic cells: Regulators of T cell development in health and HIV-1 infection. Clin Immunol 2008; 1126:1-12. Used with permission.
Antigen presentation and recognition
Antigen-presenting cells (see below) process foreign proteins into smaller peptides and express these antigens on their cell surface in association with MHC molecules, also referred to as human leukocyte antigens (HLA). T cells usually express either CD4 or CD8 molecules on their surface. These molecules are crucial in antigen recognition and in the function of the TcR. The primary function of MHC molecules is the presentation of antigen in the form of short peptides to T cells.
These molecules are essential for T cell recognition of antigens. MHC class I molecules (MHC-I) (HLA-A, B and C) are found on the surface of all nucleated cells and present antigens to CD8+ T cells. MHC-II molecules (HLA-DR, DP, DQ) are expressed only on professional antigen presenting cells such as dendritic cells (DC), B lymphocytes and macrophages. They present antigen to CD4+ T cells. Both MHC-I and MHC-II genes are highly polymorphic.
When the TcR, associated with CD3 and CD4 or CD8 molecules, binds to the antigen-MHC complex on an antigen presenting cell, cellular activation signals are transmitted from the complex of TcR/CD3. These TcR activation signals must be accompanied by co-stimulatory signals via CD28 and other co-stimulatory molecules or by cytokines. TcR signalling alone results in anergy or apoptosis. Activation of antigen presenting cells leads to expression of costimulatory molecules including CD40, CD80, CD83 and CD86. Cytokines within the inflammatory milieu and signalling from toll-like receptors (TLR), that recognise foreign pathogen associated molecular profiles, (Medzhitov, 2007; Yatim et al., 2017) activate the antigen presenting cell to enable effective co-stimulation of T cells.
Innate immune responses and the activation of adaptive immune responses
Adaptive immune responses depend on the initial recognition of pathogens. There are several pathways for recognition, including TLR 1-10, intracellular cytoplasmic sensors (including RIG-I like receptor, STING and cGAS) (Wu and Chen, 2014) and various innate lymphoid cells (ILCs). For antigen presenting cells, the initial sensing of such pathogens or cell damage is mediated by pattern recognition receptors on the dendritic cells and myeloid cells. These start the maturation and migration of the antigen processing cells as well as the upregulation of antigen processing by the DC and the upregulation and expression of MHC II on the surface of the DC. Following this, the DC can efficiently present antigens to naive T cells, which will then become effector or memory T cells. (Figure 3)
Figure 3. Innate immune responses and the initiation of adaptive immunity
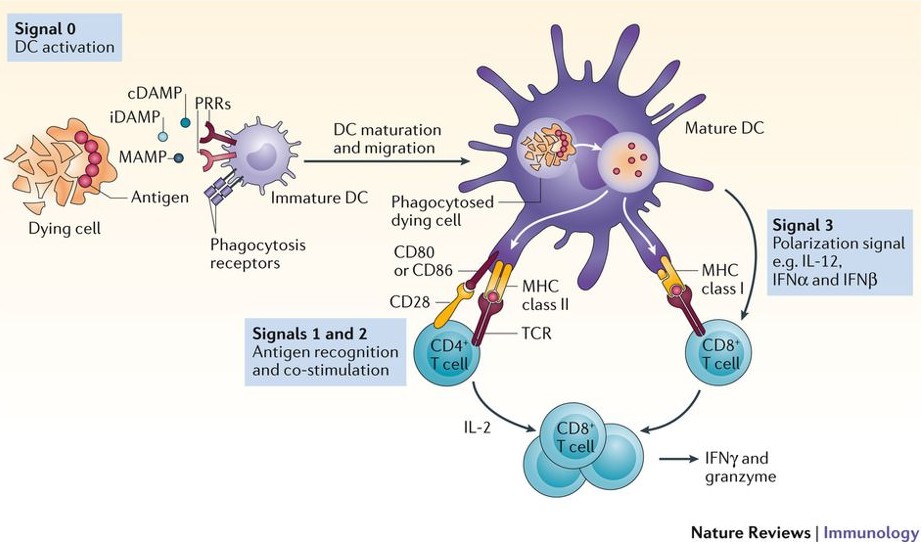
Note: The first step in the initiation of an antigen specific immune response is the maturation and migration of DCs, which is initiated by signals delivered by pattern recognition molecules and phagocytosis receptors which are part of the innate immune system. These signals, designated signal 0, can be delivered by DAMP (Danger associated molecular patterns) MAMP (Microbial associated molecular profiles) or by molecules derived from dying cells. The DC has TLRs that mediate these signals. The maturing DCs take up antigens by phagocytic receptors and C type lectin receptors, process the antigens and present them on MHC I and MHC II molecules (referred to as signal 1), as well as express costimulatory molecules (referred to as signal 2). These signals lead to stimulation of CD4+ T cells, which produce cytokines that further activate CD4+ and CD8+ T cells (signal 3 for T cell activation). The types of cytokines produced by CD4+ T cells are responsible for determining the type of T cell response that will be produced.
Source: Yatim N, Cullen S, Albert ML Dying cells actively regulate adaptive immune responses, Nature Review Immunology 2017; 17:262-275 Used with permission
T cell effector responses
Upon recognition of cognate antigens binding to the TcR expressed on the cell surface, T cells become activated and develop into effector T cells. CD4+ effector T cells function as helper cells. This help is mediated by the secretion of cytokines which influence the function of surrounding cells. CD4+ T cells can be broadly divided into sub-populations defined by the pattern of secreted cytokines and surface expression of chemokine receptors that controls their migration to lymphoid and other tissues. CD4+ T cells, in addition to broad classification as helper, cytotoxic or regulatory cells, can be described by their stage of maturation as naïve (including recent thymic emigrants), effector memory, central memory, transitional memory, terminally differentiated or stem cell memory. These stages of maturation differ in longevity and in patterns of migration, which are largely defined by their surface markers including particularly chemokine receptors and β integrins.
With cognate interactions, tnaïve T cells initially develop into non-polarised Th0 memory cells, and with subsequent stimulation develop into type 1 helper (Th1), type 2 helper (Th2) and type 17 helper (Th17) T cells, depending on cytokines present in the cellular micro-environment in which they develop.Th1 lymphocytes secrete interleukin (IL)-2 and interferon-gamma (IFN gamma), but not IL- 4, -5 or -6 whereas Th2 lymphocytes secrete IL-4, -5, -6 and -10, but not IL-2 or IFN gamma (Figure 4).
Figure 4. An overview of CD4+ and CD8+ lymphocyte responses
Need permission
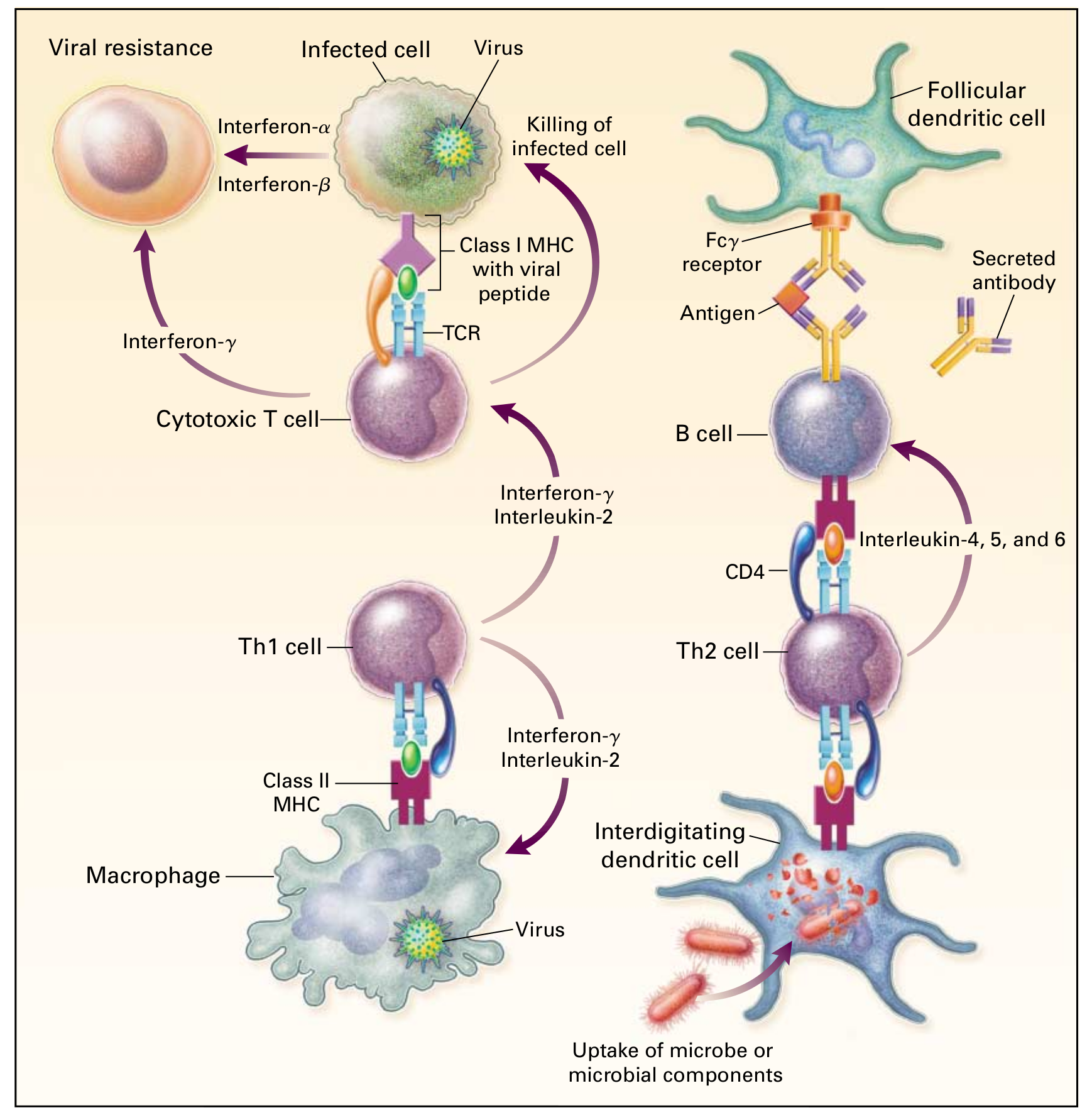
Note: T cells characteristically possess TcR that recognise processed antigen presented by MHC molecules, as shown on the left side of the figure. Most cytotoxic T cells express cell-surface CD8 and recognise processed intracellular viral antigens, which are presented as peptides by MHC-I molecules, and kill infected cells, thereby preventing or restricting viral replication. Activated cytotoxic T cells secrete interferon-gamma that, together with interferon-alpha and interferon-beta produced by the infected cells themselves, sets up a state of cellular resistance to viral infection. As shown on the right side of the figure, helper T cells are generally positive for CD4 molecules, recognise processed extracellular antigen presented by MHC-II molecules on antigen presenting cells, and can be divided into two major populations. Type 1 helper (Th1) T cells secrete interferon-gamma and interleukin-2, which activate macrophages and cytotoxic T cells to kill intracellular organisms. Type 2 helper (Th2) T cells secrete IL-4, -5, and -6, which help B cells secrete protective antibodies. B cells recognise antigen either directly or in the form of immune complexes on follicular dendritic cells in germinal centres of lymphoid tissue.
Source: Delves PJ, Roitt IM. The immune system. Second of two parts. N Engl J Med 2000; 343:108-17. All rights reserved. Used with permission.
Th1 cytokines support cell-mediated immune responses whereas Th2 cytokines generally support antibody responses. CD4+ T cells may also function as Treg cells, which develop in the thymus (central Tregs) or within the peripheral lymphoid tissue. Each of these populations has specific nuclear factors controlling their development (DuPage and Bluestone, 2016) (Figure 5).
Figure 5. Lymphocyte differentiation and diversity
Not really a better more recent overview of responses in the immunity
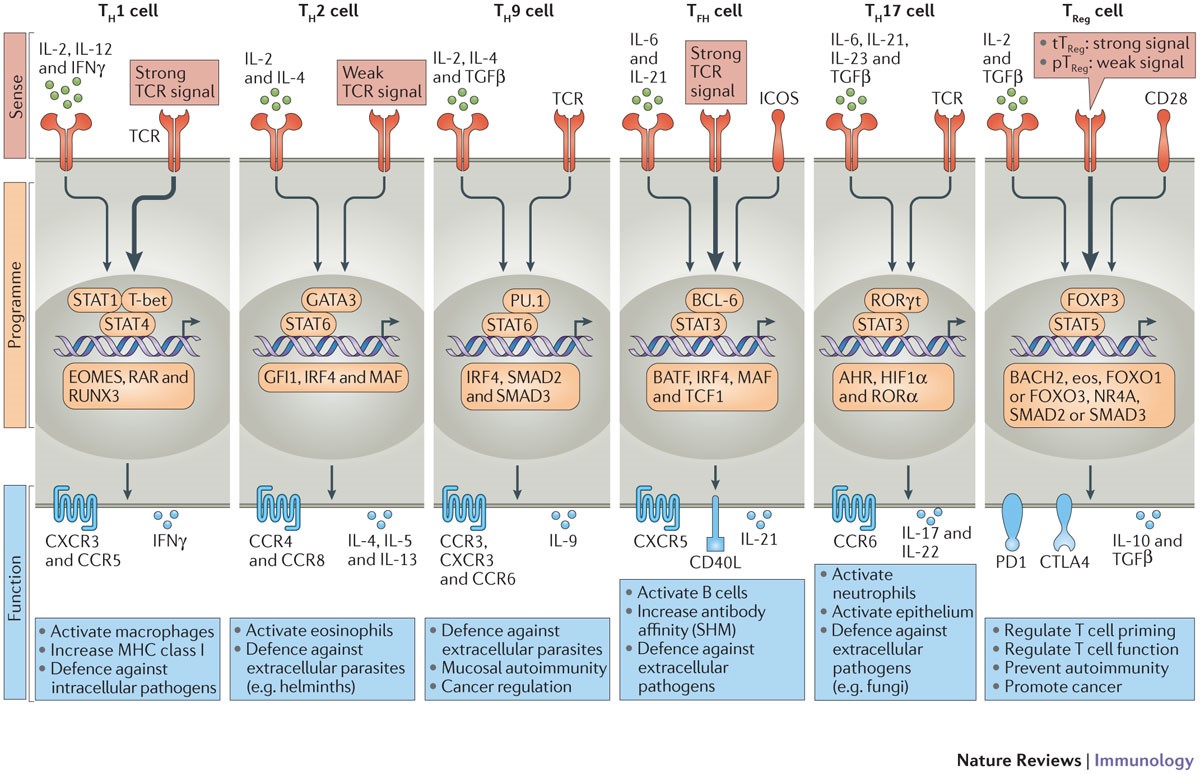
Note: Thymus-derived naïve CD4+ T cells differentiate into different sub-populations of CD4+ T cells during interaction with antigen presenting cells. Depending on the strength of TcR signalling, the particular transcription factors, the anatomical site and cytokine predominance in the micro-environment in which they encounter antigen presented by DC, they differentiate into effector or memory cells, with the phenotype determined by specific nuclear factors required for development of each subset. The effector cells express chemokine receptors that control their migration and produce specific cytokine mediators that determine their specific function.
Source: DuPage, M., and J.A. Bluestone. 2016. Harnessing the plasticity of CD4+ T cells to treat immune-mediated disease. Nat Rev Immunol. 16:149-163. Used with permission.
CD8+ T cells
CD8+ effector T cells are predominantly cytotoxic lymphocytes (CTLs). After binding to antigen on the surface of an infected cell, CTLs kill infected cells by inducing apoptosis by one of two means. Firstly, CTLs may insert perforins into the membrane of an infected cell, facilitating the passage of granzymes which promote apoptosis by direct activation of the intracellular caspase pathway. Alternatively, CTLs may bind to Fas (a protein which is expressed on the surface of the infected cell) via the Fas ligand (which is expressed on the surface of the CTL). This binding subsequently promotes apoptosis of the infected cell. CD4+ T cells also have the capacity to be CTLs under the regulation of CD8+ T cells. (Williams and Engelhard, 1997). These cytolytic CD4+ T cells induce cell death by a fas-mediated mechanism that depends on CD137 (BB-1) (Malyshkina et al., 2017). The development of CD8+ CTL requires interactions with CD4+ T cells.
CD4+ T cell memory populations
An essential feature of an adaptive immune response is the establishment of immunological memory. Each of the populations of CD4+ T cells, defined by their cytokine production and chemokine receptors (figure 5), can be further defined by the stage of development. Naïve T cells either become effector or memory T cells following their encounter with cognate antigen and DC. This occurs in the lymph node where naïve T cells entering from the blood through high endothelial venules (HEV) and encounter their cognate antigen carried from the tissue as processed antigens on antigen presenting cells. Following clonal expansion and conversion of naïve T cells to memory T cells, specific changes in the T cell phenotype ensure that the T cells recirculate into peripheral tissues and have a superior quantitative and qualitative response upon subsequent encounter with antigens.
Memory and naïve cells can be distinguished by cell surface markers. They express different isoforms of the CD45 molecule; CD45RA on naïve cells and CD45RO on memory cells, although naïve cells are more accurately defined by multiple surface markers (CCR7+, CD28+ and CD62L+). Effector memory cells re-express the CD45RA isoform but can be distinguished from naïve cells in that effector cells do not express CD62L or CCR7. Within the memory CD4+ T cell populations there are subpopulations recognised by the surface expression of specific molecules, including CD27 and the chemokine receptor CCR7, which is critical in migration of T cells into the lymph node and peripheral tissues. Cells expressing the chemokine receptor CCR6 migrate into gut and peripheral tissues whereas CXCR3 is critical for the migration and T-cell killing at sites of inflammation and virus infection. (Hickman et al., 2015)
Memory CD4+ T cells control the development of B cells and the production of high affinity antibodies. (Figure 5) Loss of CD4+ T cells is thus a critical factor in the failure of adaptive immune responses to HIV infection.
Chemokines and T cell circulation
Chemokines derive their name from being chemotactic cytokines. Their principal biological function is to regulate and direct the trafficking of cells to sites of inflammation rich in foreign antigens. Broadly, the chemokine family is divided into four subgroups: CC, CXC, C and CX3C where C represents two terminal cysteine residues and X represents the number of intervening amino acids between the two terminal cysteine residues. This structural classification also has biological significance where CXC chemokines, also referred to as alpha-chemokines, are inflammatory chemokines that attract neutrophils whereas CC chemokines, also referred to as beta-chemokines, are homeostatic chemokines principally involved in homeostatic recirculation and in attracting lymphocytes and monocytes into immune responses.
Chemokines bind to specific chemokine receptors on the surface of cells. Most of the chemokine receptors belong to the seven-transmembrane-spanning, G-protein-linked rhodopsin receptor family.(Bachelerie et al., 2014) Chemokine receptors primarily bind to chemokines within the one class of chemokines and are classified as being either CC chemokine receptors (CCR) or CXC chemokine receptors (CXCR) (Zlotnik and Yoshie, 2012). Chemokine/chemokine receptor pairs of particular significance in HIV pathogenesis are: macrophage inflammatory protein-1 alpha (MIP-1 alpha, CCL2), MIP-1beta (CCL3) and ‘regulated on activation, normal T cell expressed and secreted’ (RANTES, CCL5), which are natural ligands for CCR5 the coreceptor for HIV, and stromal derived factor-1 (SDF- 1, CXCL12), which is the natural ligand for CXCR4. Polymorphisms in these receptor/ligands have been demonstrated to influence HIV transmission and disease progression (see section on Natural history of HIV infection).
Innate lymphoid cells and NK cells
The signals for development of particular CD4+ T cell subsets depends on the TcR signal strength, antigen presenting cell function and the cytokines in the environment. There is a parallel system of development of specific innate lymphoid cells (ILCs) designated ILC1, ILC2 and ILC3 that correspond in terms of cytokine production to Th1, Th2 and Th17 CD4+ T cells. (Figure 6) (Colonna, 2018) These ILCs are predominantly tissue resident and have few if any circulating equivalents. Included among these ILC are cells that have been defined by their invariant TCR including Mucosa associated Invariant T cells (MAIT cells). These cells represent the cellular equivalent of the TLR and recognize microbial products or metabolites on antigen presenting cells. MAIT cells and ILC are lost in HIV infection. (Paquin-Proulx et al., 2017) (Mudd et al., 2018) Natural killer cells (NK cells) are also lymphocytes that mediate innate immune responses against virally-infected cells but are capable of circulating to tissues; they exist as CD56 bright KIR- IFNγ producing and a CD56 dim KIR+ cell expressing low IFNγ and the cytotoxic molecules perforin and granzyme. (Spits et al., 2016)
Figure 6. Innate lymphoid cell receptors and ligands
Need permission
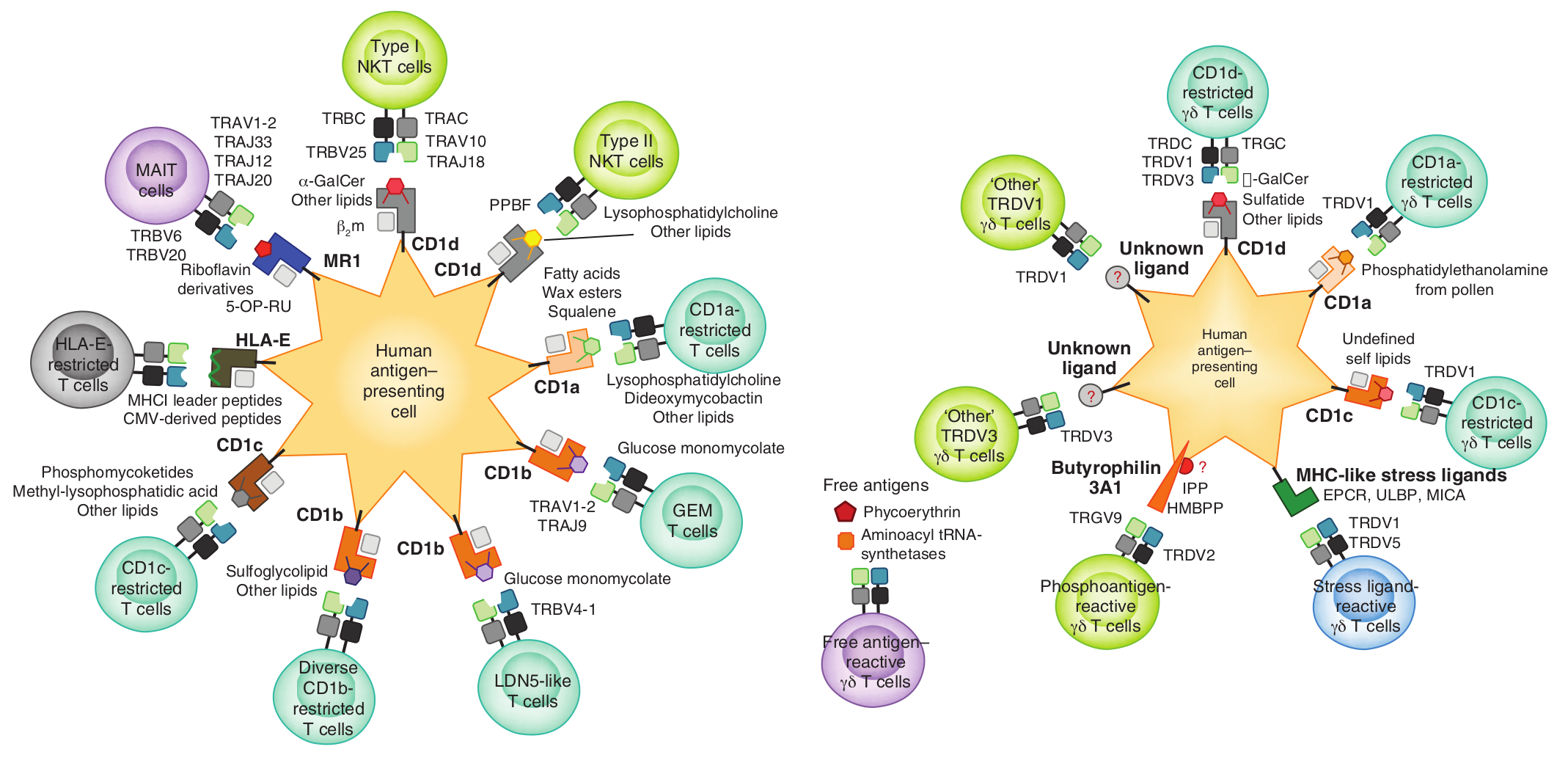
Note: A) Innate lymphoid cells are restricted by a limited range of TCRαβ and an “antigen” presented in their HLA restricting element on dendritic cells. MAIT cells are restricted by specific TCR on their surface, the MR1 molecule on antigen presenting cells and the microbial riboflavin metabolite 5-OP-RU. B) Innate lymphoid cells restricted by TCRγδ recognize a range of MHC restricted molecules with a range of ligands binding to the MHC including lipid such as α-Galactosylceramide restricted by CD1d recognized by NKT cells.
Source: Godfrey DI, Uldrich AP, McCluskey J, Rossjohn J, Moody DB. The burgeoning family of unconventional T cells. Nature Immunology. 2015 Nov; 16:1114–23. Used with permission
Lymph node architecture
The lymph node is a specialised lymphoid organ that facilitates the development of both humoral and cellular adaptive immune responses by providing a site for interaction between recirculating naïve T and B cells and antigen carrying DC migrating from tissues via afferent lymphatics. It is divided into an outer cortex and an inner medulla. The cortex contains B cell-enriched follicles that contain specialised foci called germinal centres. The germinal centres are the site of expansion of antigen-specific B cells and are involved in the generation of antibody responses, in particular the development of B cells with high affinity antigen receptors, known as B cell receptors (BCRs), which consist of immunoglobulin molecules expressed on the cell surface. This occurs through a process of affinity maturation and somatic hypermutation of immunoglobulin genes in B cells. Follicles contain DCs, follicular B lymphocytes, follicular helper CD4+ T cells (TFH) cells and a non-haematopoetic population of follicular DC (FDC) that provide a repository of immune complexes, including HIV-containing immune complexes in people with HIV infection. (Cavert et al., 1997; Estes et al., 2017) The T-cell enriched cortical regions between the follicles are known as the interfollicular regions. These thymus-dependent interfollicular regions are enriched in naïve T cells and are critical for the development of cellular immune responses. They are the site of initial activation of naïve T cells during DC-T cell interactions. The T-cell enriched regions contain antigen-presenting interdigitating DC derived directly from blood or, by migration via afferent lymphatics, from tissue. They also contain CD4+ and CD8+ T cells and some naïve B cells. The collagen matrix and fibroblastic reticular cells provide a scaffolding of the lymph node but in HIV infection there is progressive lymph node fibrosis which is a critical factor in CD4+ T cell loss (Figure 7). (Estes, 2013)
Figure 7. Lymph node architecture
Needs permission
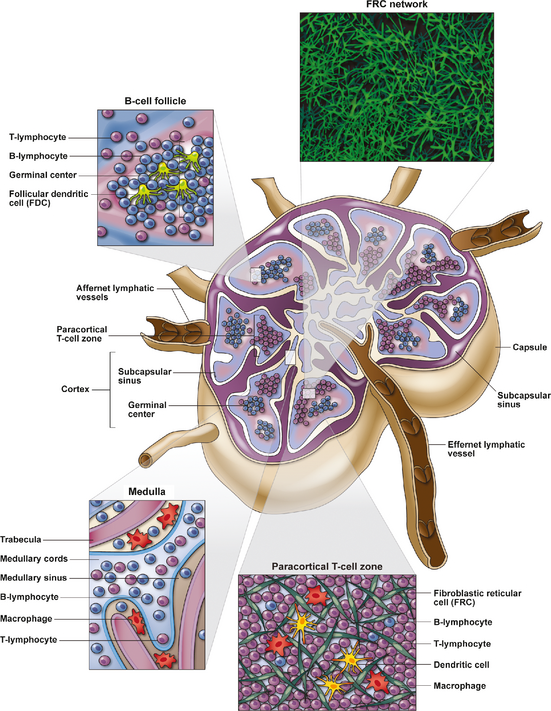
Note: Antigen migrates to the lymph node from infected tissues via the afferent lymphatics as free antigen or processed in tissue and carried by DC. Naive T cells enter from blood and interact with DC in the cortical T cell zones. B cells interact with T cells and DC in the T cell regions and then migrate into the B cell follicle where they expand to produce antigen-specific memory B cells and plasmablasts, which migrate to the bone marrow and differentiate into plasma cells and produce antibodies. FDCs ls in the germinal centres of the B cell follicle provide a reservoir of antigen/antibody immune complexes. T cells, B cells and plasmablasts migrate from the lymph node via the medulla and the efferent lymphatic vessels. Antigen-specific memory or effector B cells or T cells, activated in the lymph node, can then migrate to tissue sites of infection or inflammation. The collagen matrix and fibroblastic reticular cells network provide the scaffold of the lymphoid tissue.
Source: Estes JD. Pathobiology of HIV/SIV-associated changes in secondary lymphoid tissues. Immunol Rev 2013; 254:65–77. Used with permission.

