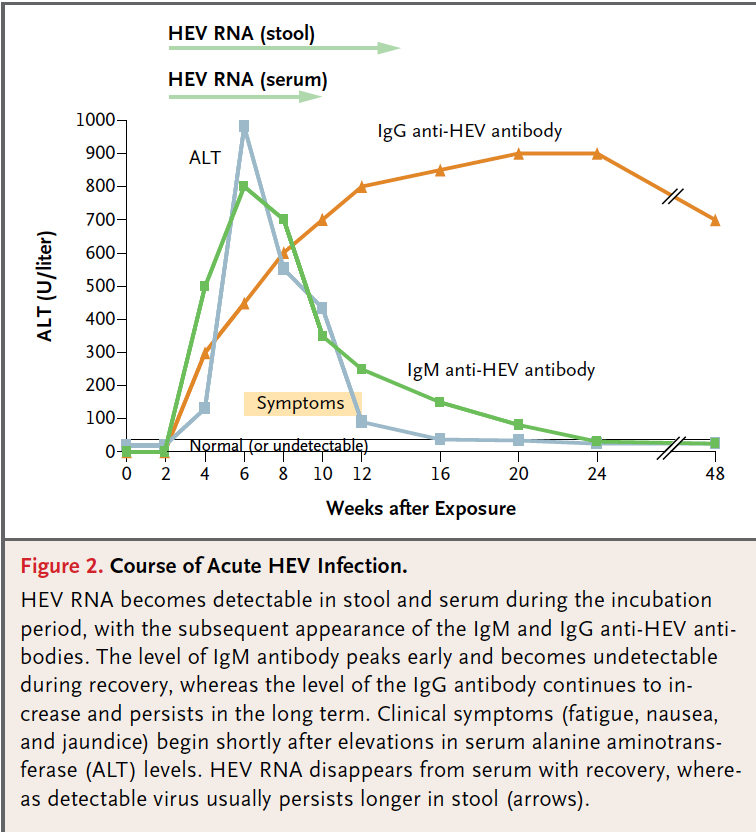
The diagnosis of HEV infection is based upon the direct detection HEV RNA in serum or stool by polymerase chain reaction (PCR) or by the detection of antibodies to HEV. Serological and nucleic acid tests (qualitative and quantitative HEV RNA) currently form the basis of HEV testing and have been used for both epidemiological and diagnostic purposes. HEV can be detected in stool during the incubation period before symptomatic illness, during the illness, and persists for as long as 2 weeks thereafter. Because HEV is enterically transmitted, patients are infectious during faecal shedding. Both IgG and IgM antibodies appear at the time of clinical onset of hepatitis, just before symptoms and elevations of liver function tests (Figure 1). Following clinical recovery, serum levels of IgG antibodies to HEV increase and usually persist lifelong while IgM antibodies wane and disappear over 3-12 months.[19] Real-time PCR is the assay of choice to detect HEV RNA and can detect all known genotypes, and is suitable for testing faecal as well as tissue samples.[20] PCR assays are still largely experimental and not widely available for routine diagnostic use.
Tests for anti-HEV antibody are commercially available but the sensitivity and specificity vary widely between assays, which probably explains the wide variation in the seroprevalence of HEV infection in different populations and the differing sensitivities between different assays in the diagnosis of PCR-proven acute HEV infection.[21] This mandates that physicians know the performance characteristics of the local assays available to them.
Figure 1. The course of acute hepatitis E infection showing the timeframes for the detection of HEV RNA, the rise of transaminases and the evolution of IgM and If G antibodies. Adapted from Hoofnagle et al. N Engl J Med 2012; 367:1237-44.[1]